Of all of the “classical” pollination syndromes, flowers that are hawkmoth pollinated have one of the highest levels of predictability. If a flower is pale in colour, opens at night, is highly scented, and possesses a long tube at the bottom of which is a supply of nectar, there’s a very high likelihood that it’s pollinated by long-tongued hawkmoths (Sphingidae).
Indeed, one of the foundational stories about the development of our understanding of how pollination systems evolve, relates to Charles Darwin, the long-tubed orchid Angraecum sesquipedale and the hawkmoth Xanthopan morganii praedicta.
Fast forward 160 years and we now know that pollination syndromes are more complex than 19th and early 20th century scientists imagined – see my recent book Pollinators & Pollination: Nature and Society for a discussion of this topic. That’s not surprising because, as I point out, we probably have data on the interactions between plants and their pollinators for only about 10% of the estimated 352,000 species of flowering plants. There’s still much to be discovered!
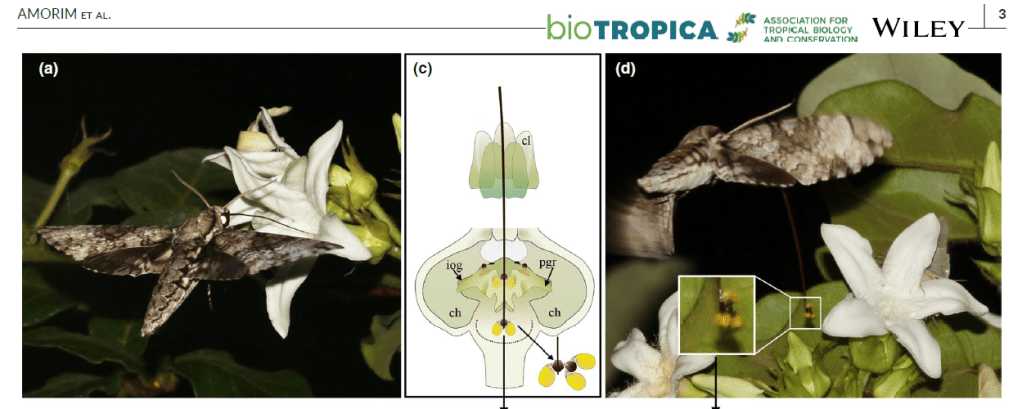
As an example of how our understanding of specialised flower-hawkmoth interactions is developing, consider this recent study that I’ve just published with my Brazilian colleague Felipe Amorim and other collaborators. In it we have shown that, contrary to expectations, a species of Apocynaceae (Schubertia grandiflora) with a relatively short floral tube can specialise on hawkmoths with much longer tongues than we might predict.
The full reference with a link to the study is shown below, followed by the abstract. If you would like a PDF, please drop me a line via my Contact page:
Abstract:
Since Darwin, very long and narrow floral tubes have been known to represent the main floral morphological feature for specialized long-tongued hawkmoth pollination. However, specialization may be driven by other contrivances instead of floral tube morphology. Asclepiads are plants with a complex floral morphology where primary hawkmoth pollination had never been described. We detailed here the intricate pollination mechanism of the South American asclepiad Schubertia grandiflora, where functional specialization on long-tongued hawkmoth pollinators occurs despite the short floral tube of this species. We studied two plant populations in the Brazilian Cerrado and recorded floral visitors using different approaches, such as light-trapped hawkmoths for pollen analysis, direct field observations, and IR motion-activated cameras. Finally, using a community-level approach we applied an ecological network analysis to identify the realized pollinator niche of S. grandiflora among the available niches in the pollinator community. Throughout a period of 17 years, long-tongued hawkmoths were consistently recorded as the main floral visitors and the only effective pollinators of S. grandiflora. Flowers rely on highly modified corona and gynostegium, and enlarged nectar chambers, to drive visitors and pollination mechanism. Despite its relative short-tube, network analysis placed S. grandiflora in the module including exclusively long-tongued hawkmoth pollinators and the most phenotypically specialized sphingophilous plants in the community. These results represent the first example of functional specialization in long-tongued hawkmoths in an asclepiad species. However, this specialization is uncoupled from the long floral tubes historically associated with the sphingophily syndrome.













